Research Progress
Research Progress
- Oxidation-responsive OEGylated poly-L-cysteine and solution properties studies
- Effects of the surface charge on the stability of PEG-b-PCL micelles: simulation of the interactions between charged micelles and plasma components
- Supramolecular Hydrogels Assembled from Nonionic Poly(ethylene glycol)-b-Polypeptide Diblock Containing OEGylated Poly-L-glutamate
- Surface-Induced Hydrogelation Inhibits Platelet Aggregation
- Stimuli-responsive polypeptide materials prepared by ring-opening polymerization of α-amino acid N-carboxyanhydrides
- Effects of molecular weight on thermal responsive property of PEGylated poly-L-glutamates
- Facile Synthesis of Dendrimers Combining aza-Michael Addition with Thiol-yne Click Chemistry
- Peptide Hydrogels Assembled from Nonionic Alkyl-polypeptide Amphiphiles Prepared by Ring-Opening Polymerization
- SYNTHESIS OF TOOTHBRUSH COPOLYPEPTIDES BASED ON POLYLYSINE BACKBONE
- One-step synthesis of water dispersible silica nanoplates
- Conformation-specific Self-assembly of Thermo-responsive Poly(ethylene glycol)-b-polypeptide Diblock Copolymer
- Coassembly of Poly(ethylene glycol)-block-Poly(glutamate sodium) and Gemini Surfactants with Different Spacer Lengths
- Thermoresponsive Oligo(ethylene glycol) Functionalized Poly‑L‑cysteine
 SYNTHESIS OF TOOTHBRUSH COPOLYPEPTIDES BASED ON POLYLYSINE BACKBONE
SYNTHESIS OF TOOTHBRUSH COPOLYPEPTIDES BASED ON POLYLYSINE BACKBONE
SYNTHESIS OF TOOTHBRUSH COPOLYPEPTIDES BASED ON POLYLYSINE BACKBONE
Lei Yu, Yu Liu, Wenxin Fu*, and Zhibo Li*
Acta Polymerica Sinica 2013, 5, 688-694
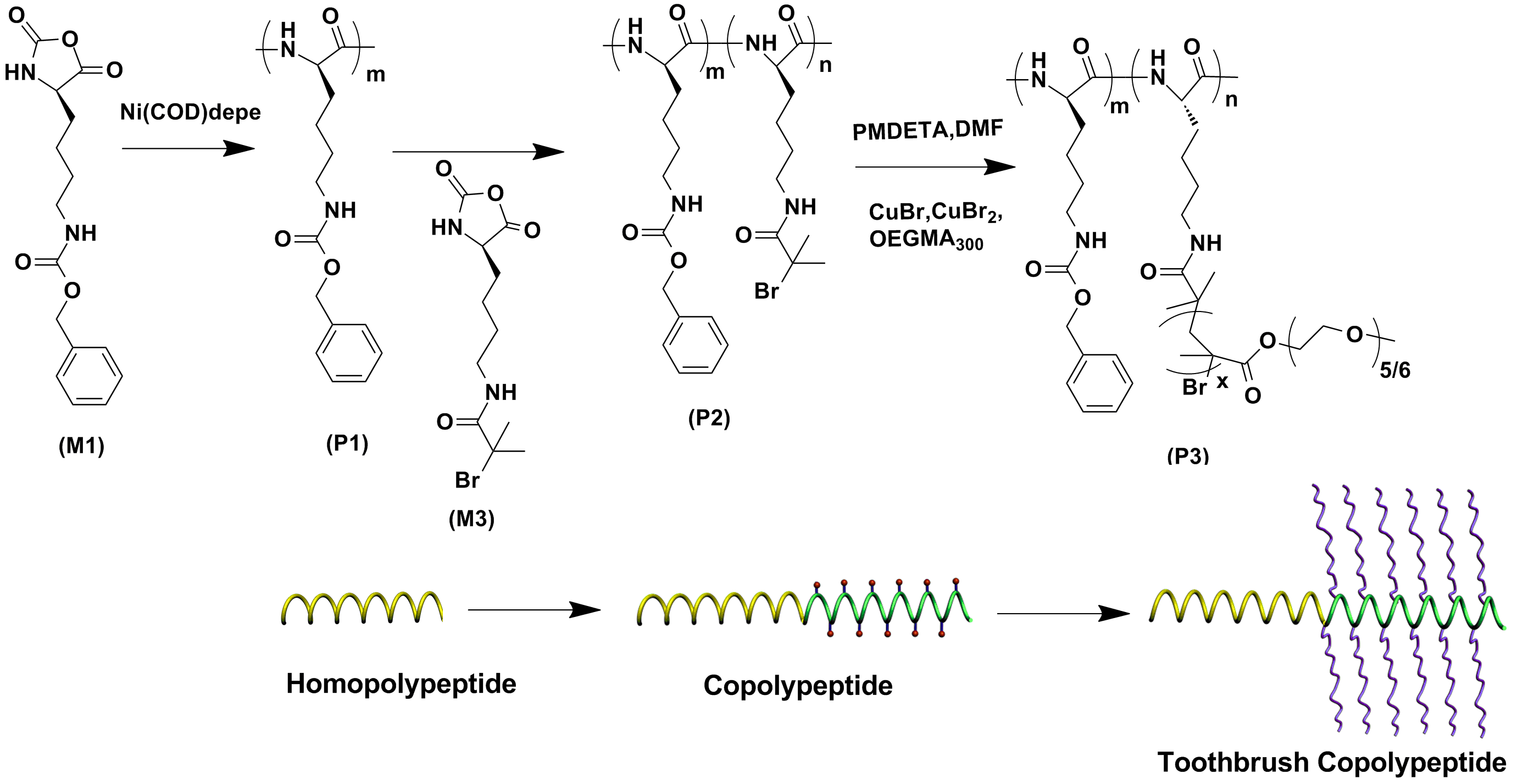
A new type of toothbrush like polypeptide with poly-L-lysine (PLL) as backbone was synthesized via a combination of ring opening polymerization (ROP) and atom transfer radical polymerization (ATRP). A Nα-bromoisobutyryl functionalized Nα-CBZ-L-lysine was prepared and subsequently converted into α-amino acid N-carboxyanhydride (Br-Lys-NCA). Then, a series of well-defined PZLL-PBrLL diblock copolypeptides were prepared via Ni(COD)depe mediated living ROP and sequential monomer addition strategy. These diblock copolypeptides containing a block of bromo-functionalized poly-L-lysine were then used as macroinitiator for subsequent ATRP of EGMA monomers to make toothbrush copolypeptides. The structure and composition of block and toothbrush copolypeptides were characterized using GPC/LLS, 1H NMR and FT-IR. The successful reaction and chain extensions were firstly supported from FTIR characterization for NCA characteristic absorbance. The GPCresults demonstrated the apparent increase of samples’ molecular weight as the linear copolypeptide was converted into copolypeptide brushes.1H NMR spectra verified the formation of homopolypeptide and copolypeptides. The secondary structures of both block and toothbrush copolypeptides were investigated using circular dichroism (CD). It was found that the α-helical content of PZLL-b-PBrLL diblock copolypeptide decreased with the reduction of mass fraction and chain length of PZLL. For toothbrush copolypeptides, the helicity was substantially lower than corresponding diblock precursors, and decreased with growing side chain length of PEGMA. Herein, we demonstrated a facile method to synthesize the biodegradable copolypeptide toothbrush with tunable spatial configurations and secondary structures by combining NCA ring opening polymerization and controlled ATRP technology. Moreover, such strategy can be easily expanded to make temperature and pH responsive copolypeptide toothbrushes.